Research Progress
Covalent organic frameworks (COFs) are the ideal templates to create metal-free catalysts because of their well-defined porous structures, predictable sites distribution and tailored environments. Attention has been put on employing different knots or linkers to advance the catalytic performance, however, the important roles of linkages toward catalyzing the oxygen reduction reaction (ORR) have not been investigated, and which linkage is more suitable for ORR is still under investigation.
Recently, a joint team lead by Prof. ZENG Gaofeng and Associate Prof. XU Qing from Shanghai Advanced Research Institute, CAS and Prof. JIANG Zheng from University of Science and Technology of China employed catalytic linkage engineering to modulate the catalytic behaviors and found the catalytic performance is determined solely by the electron states of carbon atoms in the linkages.
The research results were published in Angew Chem Int Ed.
Researchers developed catalytic COFs using different bonds such as imine, amide, azine, and oxazole bonds to link benzene units to catalyze ORR. Among these COFs, the oxazole-linkage in COFs enables to catalyze the ORR with the highest activity, which achieved a half-wave potential of 0.75V and a limited current density of 5.5mA cm -2.
Theoretical calculations showed that the carbon atoms in oxazole linkages promoted the formation of the OOH* and OH* intermediates, thus advancing the catalytic activity.
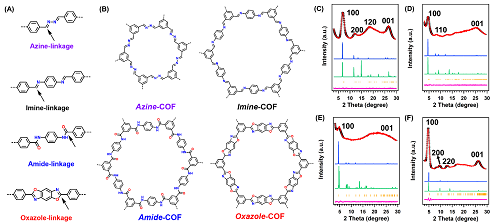
Chemical structures of COFs (azine-, imine-, amide-, and oxazole-COF) with different linkages (Image by SARI)





