Research Progress
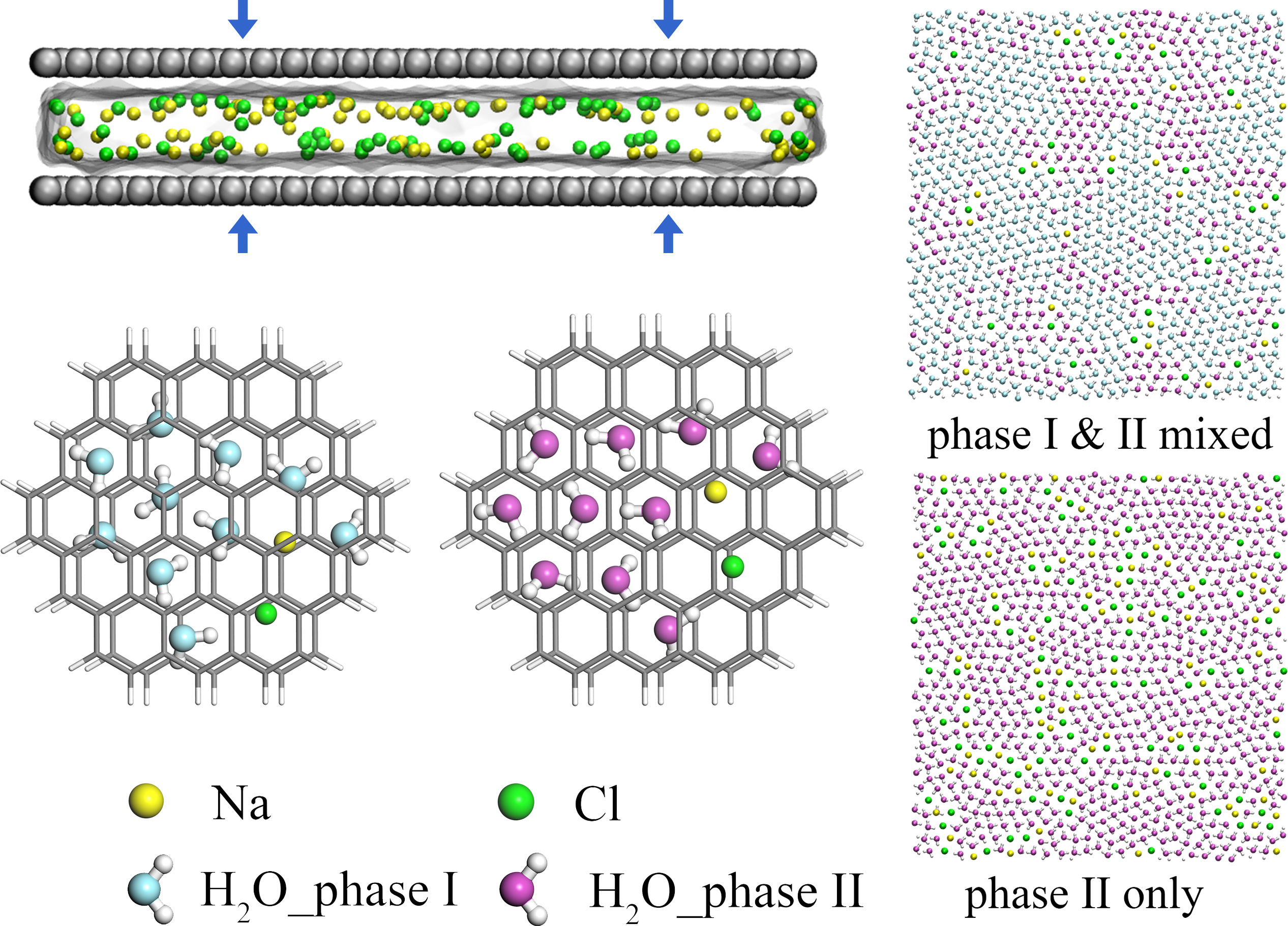
Researchers Find Novel Rhombic Ice Phase Formation from Aqueous Salt Solutions
A research team at Shanghai Advanced Research Institute (SARI) of the Chinese Academy of Sciences reported a systematic study of the two-dimensional rhombic ice formation process from various aqueous solutions at ambient temperature under strong compressed confinement of graphene.The research results were published as a Letter in Physical Review E.
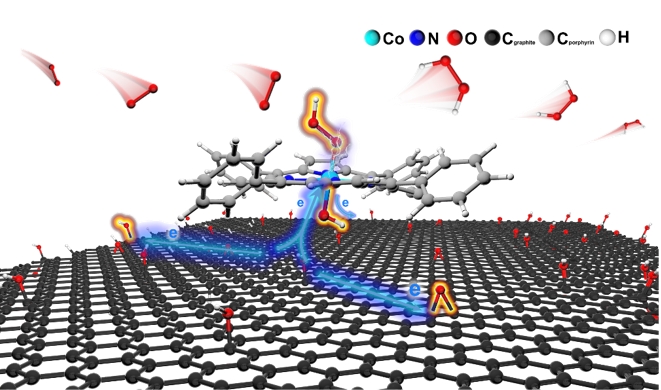
Researchers Developed Novel Molecular Electrocatalysts for Hydrogen Peroxide Electrosynthesis
A research team at Shanghai Advanced Research Institute of the Chinese Academy of Sciences proposed a strategy to promote H2O2 selectivity by designing a cobalt porphyrin supported on reduced graphene oxide molecular catalyst (CoTPP@RGO), which enabled the stable electrosynthesis of H2O2 at industrial-scale current in PEM electrolyzer. The results were published in Angewandte Chemie International Edition in June, 2024.
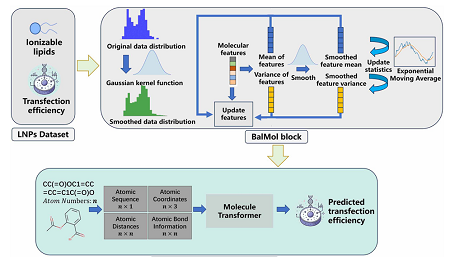
Scientists Propose a Novel Artificial Intelligence Approach for Lipid Nanoparticles Screening in mRNA Delivery
A joint research team proposed a deep learning model named TransLNP, which based on self-attention mechanisms that maps the three-dimensional microstructure and biochemical properties of mRNA-LNPs to enable high-precision automated screening of LNPs.The research findings were published in Briefings in Bioinformatics.

Chinese Scientists Call for Using Consumption-based Accounting of Carbon Emissions to Increase Fairness
A new study by Chinese scientists, released on May 29 in Shanghai, has called for the use of consumption-based accounting (“CBA”) emissions in calculating global carbon emissions in order to help make allocating responsibility for reducing emissions just and fair.The study,"Research Report on Consumption-based Carbon Emissions (2024)" ("the Report"), was jointly completed by scientists from Shanghai Advanced Research Institute, CAS, University of Chinese Academy of Sciences, Institute of Urban Environment, CAS and Tsinghua University.
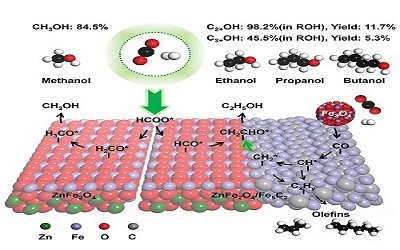
Scientists Discover Novel Strategy for Selectivity Tuning in Alcohol Synthesis by Crystal Phase Engineering
A research team at Shanghai Advanced Research Institute (SARI) of the Chinese Academy of Sciences reported for the first time an efficient FeZn-based catalyst for methanol synthesis via CO2 hydrogenation and by crystal structure, achieving high methanol selectivity of 84.5%.The research results were published in the latest issue of Chem.
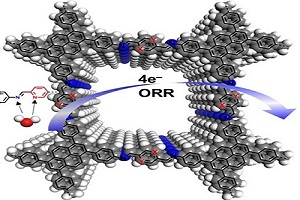
Researcher Propose Solvent Effects on Metal-free Covalent Organic Frameworks in Oxygen Reduction Reaction
A research group at Shanghai Advanced Research Institute (SARI) of the Chinese Academy of Sciences, represents the first investigation into the solvent effect on COFs for catalyzing the oxygen reduction reaction (ORR). They found all COFs synthesized with different nitrogen atoms exhibited good crystallinity and high surface areas, but displayed different binding abilities towards water molecules.This work was published in Angew. Chem. In. Ed.





